
The One Maintenance Task That Most Golf Cart Owners Get Wrong
Of all the maintenance tasks that keep a flooded lead-acid golf cart battery healthy, watering is both the simplest and the most commonly botched. Not because the task itself is difficult — you add water to the cells until they reach the correct level. That is genuinely the whole procedure. But in practice, owners make three specific mistakes that accelerate battery degradation in ways that are silent, cumulative, and completely preventable.
Mistake one: using tap water instead of distilled water. Tap water contains dissolved minerals — calcium, magnesium, iron, chlorides, and, depending on your water supply, other contaminants — that precipitate onto the battery plates when the water evaporates during charging. These deposits reduce the active surface area of the plates, increase internal resistance, and accelerate degradation. Every gallon of tap water used in a golf cart battery contains enough dissolved solids to cause measurable damage.
Mistake two: adding water before charging instead of after. The electrolyte level in a flooded lead-acid cell rises as the battery charges and falls as it discharges. If you add water to a discharged battery and then charge it, the electrolyte expansion during charging will push the now-overfilled electrolyte out through the cell caps, spilling sulfuric acid onto the battery terminals, cables, and battery tray. Filling after charging is the correct sequence — not before.
Mistake three: not watering at all. This is the most common failure mode, and it produces the most serious damage. A cell with the electrolyte level below the plate tops has plates exposed to air. Lead plates in air oxidize irreversibly. Even a few days of exposure causes permanent plate surface damage that no amount of watering afterward can reverse.
This guide covers the chemistry behind all three of these mistakes in detail, gives you the complete correct watering procedure, tells you what to look for when inspecting your cells, and answers every question about distilled water — including whether filtered water, RO water, or bottled water is an acceptable substitute.
The Chemistry: What Is Actually in Your Golf Cart Battery’s Electrolyte

The electrolyte in a flooded lead-acid golf cart battery is a solution of sulfuric acid (H₂SO₄) and water (H₂O). The sulfuric acid provides the ionic medium through which electrical current flows between the plates—it is not a passive liquid but an active participant in the energy storage chemistry. The water is not a filler—it is chemically consumed and regenerated during every charge-discharge cycle.
During discharge, the sulfuric acid reacts with the lead plates to form lead sulfate, releasing electrons that power your cart’s motor. The concentration of sulfuric acid in the electrolyte decreases during this process. During charging, the reverse reaction occurs — lead sulfate converts back to lead dioxide and sponge lead, releasing sulfuric acid back into the electrolyte. The acid concentration rises as the battery charges.
The water component participates in a different way. During charging, particularly during the final stages of charging when the battery approaches full capacity, some of the current electrolyses water molecules into hydrogen gas (at the negative plate) and oxygen gas (at the positive plate). These gases bubble through the electrolyte and vent through the cell caps. This is the normal gassing that you can see and faintly smell during charging—it is not a sign of a problem; it is the normal end-of-charge process. But it consumes water from the electrolyte, which is why the water level gradually drops and must be periodically replenished.
Why Water Purity Is the Critical Variable
Pure water — that is, water containing only hydrogen and oxygen atoms with no dissolved minerals, salts, or organic compounds — participates in the battery chemistry without introducing any contaminants. When it is electrolyzed during charging, only hydrogen and oxygen are produced. When it evaporates or gasses off, nothing is left behind.
Impure water introduces dissolved substances that do not participate in the lead-acid chemistry and cannot be vented off. When the water is consumed through electrolysis and evaporation during charging, these dissolved impurities remain in the cell and concentrate over time. Eventually, they deposit on the plate surfaces, contaminate the electrolyte chemistry, or create competing electrochemical reactions that reduce battery performance.
The specific contaminants found in typical tap water and their effects on battery chemistry:
| Contaminant | Typical Tap Water Level | Battery Impact | Mechanism of Damage |
| Calcium (Ca²⁺) | 20-200 ppm | HIGH | Calcium sulfate (CaSO₄, gypsum) forms when calcium contacts the sulfuric acid electrolyte. Deposits on plate surfaces, increasing internal resistance and reducing active material participation. Insoluble — cannot be removed once deposited. |
| Chloride (Cl⁻) | 10-250 ppm | VERY HIGH | Chlorides react with lead to form lead chloride, which is highly soluble and disrupts the passivation layers on the plates. Accelerates grid corrosion and positive plate degradation. Even small amounts are harmful — chloride levels as low as 5 ppm cause measurable capacity loss over time. |
| Iron (Fe²⁺/Fe³⁺) | 0.01-3 ppm | HIGH | Iron ions act as a catalytic contaminant. They migrate between positive and negative plates, causing a self-discharge reaction that prevents the battery from holding charge. Even 1 ppm iron contamination measurably increases the self-discharge rate. |
| Magnesium (Mg²⁺) | 5-50 ppm | MODERATE | Magnesium sulfate is more soluble than calcium sulfate but still deposits on plates at higher concentrations. Less damaging than calcium or chloride but contributes to long-term capacity reduction. |
| Copper (Cu²⁺) | 0-2 ppm | VERY HIGH | Copper ions deposit as copper metal on the negative plate during charging, causing localized corrosion and blocking active material sites. Even trace amounts are significant. Not common in municipal water but occurs in some well water and older copper piping. |
| Chloramine/Fluoride | Varies | MODERATE | Water treatment additives. Chloramine partially decomposes to chloride in the acidic electrolyte—contributing to the chloride damage described above. Fluoride can react with lead to form lead fluoride deposits. |
| Total Dissolved Solids (TDS overall) | 50-500 ppm (typical tap water) | HIGH | Battery-grade distilled water should measure below 1 ppm TDS. Typical tap water is 50-500 ppm. Every fill with tap water introduces 50-500 times the acceptable contaminant level. |
Distilled Water vs Every Alternative: What Is Actually Acceptable

The distilled water question generates a surprising amount of confusion online, with people claiming that filtered water, bottled water, or reverse osmosis water is equivalent to distilled. Some of these claims are closer to correct than others. Here is the definitive breakdown.
True Distilled Water: The Standard
Distilled water is produced by boiling water to steam and then condensing that steam in a separate vessel. Minerals, salts, and most contaminants do not evaporate with the water — they remain behind in the boiling chamber. The condensed steam is the distilled water: essentially pure H₂O with typical TDS measurements of 0.5-1 ppm.
Battery-grade distilled water is specifically tested and certified to meet ASTM D1193 Type II specifications, which requires total dissolved solids below 0.1 ppm and specific limits on iron, chloride, and other key contaminants. Most distilled water sold in gallon jugs at grocery stores or automotive retailers meets these specifications, though they are rarely explicitly labeled as battery grade.
The practical test: a TDS meter (available for $10-$15 online) measures the dissolved solid content of any water sample. Battery-appropriate water should read 0-5 ppm on a TDS meter. Tap water from most US municipal supplies reads 50-300 ppm on the same meter — a striking visual demonstration of why the two are not interchangeable.
Reverse Osmosis (RO) Water: Acceptable with Verification
Reverse osmosis filtration forces water through a semipermeable membrane that removes most dissolved ions. Well-maintained RO systems typically produce water in the 10-50 ppm TDS range—significantly better than tap water but potentially 10-50 times higher TDS than distilled water.
RO water is an acceptable battery watering option if you verify the actual TDS output with a meter. An RO system producing 5-10 ppm TDS water is acceptable for battery use. An RO system that has not had its membrane replaced in two years and is producing 80-100 ppm water is not efficient. RO membrane performance degrades over time and with water quality — a meter reading gives you the actual current output, not the rated performance when the system was new.
The additional caveat: RO systems do not remove all contaminants equally well. Some chlorine compounds and small organic molecules pass through RO membranes more readily than mineral ions. For a battery application where even trace chloride contamination is significant, distilled water’s additional purification step makes it the more reliable choice.
Filtered Water (Brita, PUR, etc.): Not Acceptable
Consumer water filters using activated carbon remove chlorine compounds, organic matter, and some heavy metals. They do not remove dissolved mineral ions—calcium, magnesium, sodium, and potassium pass through carbon filters essentially unchanged. A Brita-filtered tap water sample will typically measure 40-200 ppm TDS, depending on the source water. This is not meaningfully better than unfiltered tap water for battery purposes.
If a filter is connected to a tap that supplies very low-TDS well water or municipal water, the filtered output may happen to be low-TDS—but this is a coincidence of the source water quality, not a property of the filtration. Always measure with a TDS meter rather than assuming filter type determines suitability.
Bottled Spring Water: Absolutely Not
Spring water is specifically marketed for its mineral content — the calcium, magnesium, and bicarbonate that give it a pleasant taste and make it perceived as healthier than distilled water. These are exactly the contaminants that damage battery plates. Spring water TDS levels typically run 200-500 ppm. It is essentially the worst possible alternative to distilled water for battery use, despite being commonly suggested in uninformed sources.
Deionised Water: Equivalent to Distilled
Deionized water is produced through an ion exchange process that replaces mineral cations (calcium, magnesium, and sodium) with hydrogen ions and mineral anions (chloride and sulfate) with hydroxide ions—which then combine to form water. The result is water with extremely low TDS, typically 0.1-1 ppm, equivalent to or better than distilled water for battery purposes.
Deionized water is less commonly available at retail than distilled but is used in laboratory and industrial settings. If you have access to deionized water from a verified source, it is an acceptable and equivalent substitute for distilled water in golf cart batteries.
| Water Type | Typical TDS (ppm) | Battery Suitability | Notes |
| Battery-grade distilled | < 1 ppm | EXCELLENT | The gold standard. ASTM D1193 Type II certified. Available at grocery stores and auto parts retailers. |
| Deionised (DI) water | 0.1 – 1 ppm | EXCELLENT | Equivalent to distilled. Used in labs and industrial settings. Less commonly available at retail. |
| RO water (verified < 10 ppm) | 5 – 50 ppm (varies) | ACCEPTABLE (verify TDS) | Only acceptable if TDS is verified below 10 ppm with a meter. RO system performance degrades—always measure, never assume. |
| Distilled from home unit | < 5 ppm | GOOD | Countertop distillers produce acceptable water if operated correctly and vessels are clean. Verify with a TDS meter initially. |
| Filtered tap water (Brita, etc.) | 40 – 200 ppm | NOT ACCEPTABLE | Carbon filters remove chlorine and organics but not mineral ions. TDS barely reduced from the source water. |
| Municipal tap water | 50 – 300 ppm | NOT ACCEPTABLE | Theoretically close to distilled, but picks up atmospheric contaminants and collection vessel contamination. It’s only acceptable if TDS is verified below 5 ppm from a clean collection system. |
| Well water | 100 – 800 ppm | NOT ACCEPTABLE | Contains calcium, magnesium, and chlorides. All cause cumulative plate contamination. Never use in golf cart batteries. |
| Spring / mineral water | 200 – 500 ppm | NOT ACCEPTABLE | Specifically marketed for mineral content — the exact contaminants that damage battery plates. Never use. |
| Rainwater (collected) | Varies widely | RISKY | Theoretically close to distilled but picks up atmospheric contaminants and collection vessel contamination. It’s only acceptable if TDS is verified below 5 ppm from a clean collection system. |
The Complete Correct Watering Procedure: Step by Step

Now that you have the right water, the procedure matters almost as much as the water type. Here is the correct sequence—including the timing, tools, and safety requirements that most guides either gloss over or omit entirely.
Safety Requirements Before You Start
Flooded lead-acid batteries contain sulfuric acid electrolyte and produce hydrogen gas during and after charging. Before opening any cell cap:
- Wear safety glasses. Electrolyte splash from a pressurized cell, a spill, or an accidental contact produces immediate and serious eye injury. Safety glasses are not optional for battery work.
- Wear nitrile or rubber gloves. Electrolyte spilled on skin causes chemical burns that are initially mild but worsen if not immediately flushed out. Diluted electrolyte on hands feels like nothing—and is still causing tissue damage.
- Ensure the battery area is well-ventilated. Charged batteries continue to release small amounts of hydrogen gas for 1-2 hours after a charge cycle ends. Perform watering in open air or a well-ventilated garage — not a sealed room.
- Keep sources of ignition away from the battery area. Hydrogen is explosive at concentrations above 4% in air. An enclosed garage with recently charged batteries and a gas water heater pilot light is a genuine hazard.
The Complete Watering Procedure
- This is the most important timing rule. The electrolyte level rises as the battery charges—if you check levels on a discharged battery, the level appears lower than it actually is after charging. Filling a discharged battery and then charging it produces overfilling, which pushes electrolyte out of the cell and causes corrosion, terminal damage, and potential acid spills. Complete a full charge cycle first.
- Wait 30-60 minutes after the charger shuts off before checking levels. This allows the electrolyte to settle and the temperature to return toward ambient. Hot batteries have expanded electrolyte that gives a misleadingly high level reading. Allow the battery to rest after charging.
- You will need distilled water, a battery watering gun or large syringe with a flexible tip (a turkey baster works), a flashlight or headlamp, safety glasses and gloves, and a dry cloth for any spills. Gather your tools.
- Most golf cart batteries have individual vent caps on each of the six cells. Some newer batteries have a single multi-cell cap assembly. Remove caps gently — do not force caps that feel stuck, as excessive force can crack the battery case. Remove the cell caps one at a time.
- Shine the light into the open cell. You should be able to see the top of the electrolyte level below the bottom of the filler tube. The correct fill target is when the electrolyte level is between 3/8 and 1/2 inch below the bottom of the filler tube—you will see the tube end, then a gap of air, then the electrolyte surface below. Check the electrolyte level with the flashlight.
- Use the watering can or syringe to add water slowly. Do not pour directly from a bottle — you will overshoot the level quickly. Add a small amount, check the level, and add more if needed. The goal is the electrolyte surface sitting comfortably below the filler tube bottom—not touching it, not several inches below it. Add distilled water to reach the correct level.
- This is the mistake that causes terminal corrosion and acid spills. Overfilled cells push electrolyte out during the next charge cycle. When electrolyte reaches the terminals and battery tray, it causes corrosion that is much more destructive than simply having low water levels. Never overfill.
- Press each cap down until it snaps or seats securely. Loose caps allow electrolyte spray to escape during charging and reduce the effectiveness of the pressure relief valve that prevents overpressure. Replace the cell caps firmly.
- A dry cloth for water. Baking soda solution (1 tablespoon in 1 cup of water) for any electrolyte spills—the baking soda neutralizes the acid before it causes corrosion. Clean any spilled water or electrolyte immediately.
- Set a 30-45 day reminder for the next check. Seasonal adjustment: hot summer months increase water consumption significantly — check every 2 weeks in temperatures above 90°F during heavy use. Record the date.
Water Consumption: What Is Normal and What Is a Warning Sign
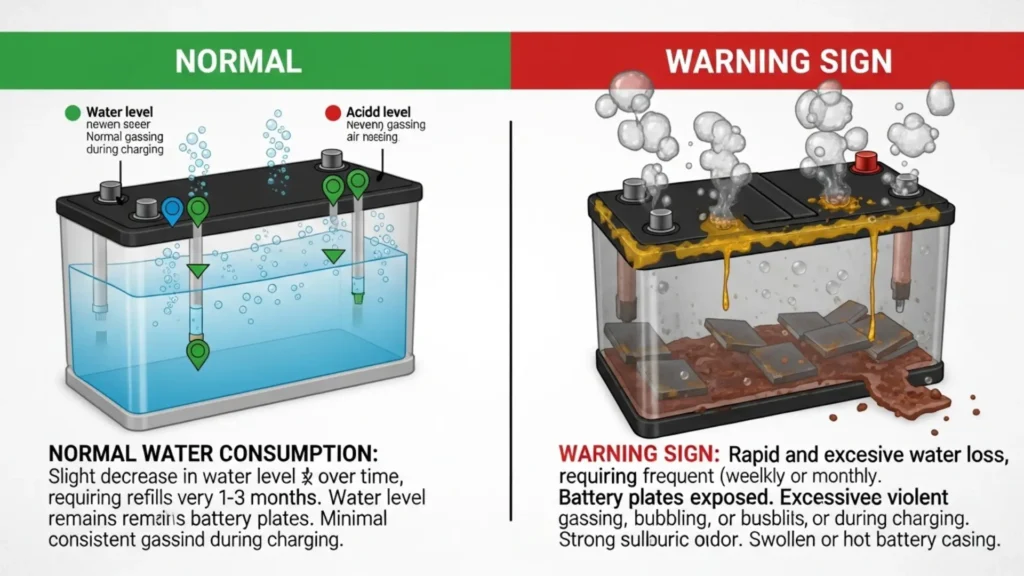
Golf cart batteries consume water at a predictable rate that varies with use intensity, charging frequency, temperature, and battery age. Understanding what constitutes normal water consumption helps you identify early warning signs before they become significant problems.
Normal Water Consumption Ranges
A regularly used 48-volt cart (charged daily or near-daily) on a healthy pack with a properly calibrated charger typically adds approximately 1-4 ounces of distilled water per battery per month during moderate climate conditions. On a six-battery pack, that is 6-24 ounces (roughly one third to one and a half cups) per month across the entire pack.
This seems like a small amount—and it is. Properly maintained batteries with a correctly calibrated charger do not guzzle water. Heavy water consumption is often a sign of a charger problem, not a sign that the batteries need frequent watering.
| Condition | Expected Water Consumption | Check Interval | Notes |
| Normal use, quality charger, 65-80°F temperature | 1-3 oz per battery per month | Monthly | Baseline for a well-maintained cart in a mild climate. Charger terminates cleanly at correct absorption voltage. |
| Heavy use (daily + hilly terrain), mild temperatures | 2-5 oz per battery per month | Every 3 weeks | Heavy cycling increases gassing during charging. Higher current draw = more charging cycles = more water loss. |
| Hot climate (85-100°F+ ambient), normal use | 3-8 oz per battery per month | Every 2-3 weeks | Heat accelerates both electrolysis and evaporation. Summer months in Florida, Texas, and Arizona require more frequent checks. |
| Overcharging / charger not terminating correctly | 8-20+ oz per battery per month | Weekly | WARNING: Excessive water consumption indicates chronic overcharging. Check charger absorption voltage. A charger that does not terminate correctly is boiling off electrolyte and shortening battery life. |
| Battery with shorted or failing cell | Very high in one cell only | Check immediately | One cell consuming significantly more water than others is a classic symptom of an internal short in that cell. Have the battery load tested immediately. |
The key diagnostic insight from this table: if you are adding large amounts of water every month—more than a cup per battery—and your charger is not ancient, the charger is almost certainly the source of the problem. A charger that does not terminate cleanly at the correct absorption voltage continues pushing current into a fully charged battery, electrolyzing water at a high rate. The batteries are not consuming water abnormally — the charger is forcing abnormal gassing. Address the charger before continuing to invest in water and maintenance on a pack that is being slowly overcharged to death.
What You See in the Cells: Interpreting Electrolyte Appearance and Level
When you open the cell caps and look inside, the electrolyte gives you information beyond just the level. Here is how to read what you are seeing.
Electrolyte Colour
Healthy electrolyte is clear to very slightly amber—essentially colorless when fresh and developing a very faint golden tint after years of service. Dark brown or dark amber electrolyte indicates significant plate shedding has occurred—the brown color comes from fine lead dioxide particles suspended in the electrolyte. This is a sign of an aging battery that has been through many charge cycles, but it is not an immediate emergency unless accompanied by performance problems.
A milky or cloudy white electrolyte indicates contamination—either an additive has been introduced, or the battery has been filled with hard water that has deposited calcium sulfate in the electrolyte. This is a more concerning sign and suggests the battery may have been damaged by contaminated water at some point in its history.
Very dark or black electrolyte with visible sediment at the cell bottom indicates severe plate shedding. This is a late-stage battery that has shed significant active material. The sediment at the bottom of the cell may be approaching the plate level, risking an internal short. This battery is near the end of its life regardless of water or charging intervention.
Electrolyte Level Situations
Electrolyte level significantly below the plates: This is an emergency situation. Exposed plates are oxidizing right now. Add distilled water immediately to cover the plates, but understand that any oxidation that has already occurred on the exposed sections is permanent. The degree of recovery depends on how long the plates were exposed—hours versus days versus weeks produces very different outcomes.
Electrolyte level is low, but plates are still covered: Normal maintenance situation. Add distilled water after a full charge to bring the level to the correct position. Check the other cells in the pack—if one cell is consistently lower than others, that cell may have a small crack or higher consumption rate, requiring more frequent monitoring.
Electrolyte level above the filler tube bottom: Overfilled. This typically means water was added to a discharged battery, and the level rose above the correct point when the battery was subsequently charged. If the overfill is modest (just touching the filler tube bottom), the next charge cycle may push some electrolyte out. Place absorbent material around the batteries for the next charge and clean up afterward. Severe overfill where electrolyte is visible at the cap level should be carefully siphoned back to the correct level using a bulb syringe.
Measuring Electrolyte with a Hydrometer: The Deeper Diagnostic Tool

A battery hydrometer measures the specific gravity of the electrolyte — the density of the sulfuric acid solution relative to water. Since the acid concentration changes with the battery’s state of charge (higher concentration = more charged), specific gravity is a direct measure of the battery’s charge state and a useful indicator of cell health.
Fully charged flooded lead-acid golf cart cells should measure 1.265-1.280 specific gravity (at 77°F / 25°C reference temperature). A fully discharged cell measures approximately 1.120-1.150 V. Most cells in a healthy, fully charged pack will read within 0.010-0.015 of each other—tight uniformity indicates balanced cell health.
What the hydrometer reveals that a voltmeter does not: a cell with a specific gravity reading significantly lower than others after a full charge—say, 1.215 when others read 1.265—indicates that this cell is not accepting charge normally. This could be severe sulfation on the plates, internal plate damage, or a slight internal short. The specific gravity variation between cells is the earliest indicator of a developing problem, appearing months before the problem becomes visible as a voltage drop or performance loss.
Temperature correction is required for accurate hydrometer readings. The standard reference temperature is 77°F. For every 10°F above 77°F, add 0.004 to the reading. For every 10°F below 77°F, subtract 0.004. A cell reading 1.255 at 100°F actually has a corrected specific gravity of 1.265—right in the healthy range. The same reading at 50°F corrects to 1.244 — slightly below ideal, suggesting incomplete charging.
Seasonal Adjustments: Hot Weather, Cold Weather, and Storage
Hot Weather — Increase Check Frequency
Summer heat is the most demanding season for electrolyte management. Every 15°F increase in ambient temperature approximately doubles the rate of water consumption from electrolysis and evaporation. A cart stored in a garage in Phoenix, Arizona, in July, where temperatures reach 110°F, is losing water at four or more times the rate of the same cart in a 75°F spring climate.
In hot climates during summer months, check electrolyte levels every two weeks rather than monthly. Keep a gallon of distilled water accessible near the cart for easy refills. Never let the pack go more than three weeks without a level check during peak summer heat—the consequences of running a cell dry in 105-degree heat are permanent plate damage that shortens the pack’s life.
Cold Weather — Less Frequent But Still Required
Cold temperatures reduce water consumption from electrolysis and evaporation, so winter months require less frequent watering than summer. However, cold also affects electrolyte behavior in a way that creates a watering trap: cold, discharged batteries have very low electrolyte levels that rise significantly when the battery is charged and warmed. Adding water to a cold, discharged battery and then charging it produces overfilling.
In cold weather, the same rule applies as year-round: always check and water after a full charge, never before. The temperature-related electrolyte expansion is even more pronounced in cold weather, making the timing of the inspection more important.
Winter Storage — The Pre-Storage and Post-Storage Checklist
For carts stored through winter, a specific watering protocol applies at both ends of the storage period. Before storage: perform a full charge and equalization, then check and correct all electrolyte levels. A fully charged battery has slightly higher electrolyte that has been well-mixed by the charging process—this is the correct state to inspect from. After storage: before the first charge of spring, inspect electrolyte levels and top up if any cell has dropped during storage self-discharge. Then perform a full charge before use.
The reason for the post-storage check: even without charging, batteries self-discharge slowly during storage. The sulfation that occurs during storage consumes some water and slightly lowers the electrolyte level. Starting the spring season with a watering check and full charge gives the pack the best chance of recovering from any storage degradation.
Buying Distilled Water: Where to Get It and How Much You Need

Distilled water for golf cart batteries is widely available and inexpensive. The question most owners have is how much to buy and where to get it consistently.
Volume estimate: for a standard six-battery 48V pack under normal conditions, approximately half a gallon to one gallon per month covers routine maintenance. Buying two one-gallon jugs every two months is a reasonable stock for most residential owners. In summer months in hot climates, doubling that estimate is prudent.
Sources for battery-grade distilled water:
- Grocery stores: the most common and convenient source. Distilled water is typically sold in one-gallon jugs in the water aisle. Most grocery store distilled water meets battery-grade specifications despite not being labelled as such.
- Auto parts stores (AutoZone, O’Reilly, Advance Auto Parts): explicitly stock battery-grade distilled water, often in clearly labeled battery maintenance jugs. Marginally more expensive than grocery store distilled, but guaranteed to the correct specification.
- Golf Cart Gears: Battery watering kits, including distilled water, watering guns, and hydrometers, bundled for convenient ordering.
- Warehouse stores (Costco, Sam’s Club): larger volume purchasing at a lower per-gallon cost. If you have a large fleet or multiple carts, warehouse purchasing reduces per-unit cost.
Storage: Distilled water can be stored for extended periods in its sealed original container. Once opened, store in a clean, sealed container away from contaminants. Avoid storing it in the same location as cleaning chemicals—airborne contaminants from open chemical containers can affect the water quality over time.
What We See When Customers Call About Water-Related Battery Problems

Two water-related patterns generate the most service calls and the most preventable battery replacement costs on our support line.
The first is the tap water addition. An owner calls because their 2-year-old batteries are performing like 4-year-old batteries—the range has dropped significantly, and the pack feels weak on hills. When we ask what water they have been using, the answer is often, ‘Just regular tap water—is that a problem?’ At this point, it is a problem that cannot be reversed. The contamination is on the plates, and the capacity loss is permanent. The batteries are not ruined — they will continue working — but they will continue degrading faster than they should have been. The lesson for every other cart owner: there is no second chance on this one. The damage from two years of tap water cannot be undone. Use distilled water from day one.
The second is the overfill cascade. An owner topped up the batteries before a charge; the charger pushed electrolyte out during charging, the electrolyte reached the terminals, and it began corroding the battery cables. A month later, the cart starts performing poorly — not because the batteries are dying but because the corroded cable connections are creating resistance in the pack circuit that drops voltage under load. The owner assumes it is a battery problem. It is a cable problem caused by an overfill that caused a spill that caused corrosion. The fix is cable replacement and terminal cleaning — but none of it would have happened if the filling sequence (after charge, not before) had been followed.
Both of these are completely preventable with the information in this guide. The investment in correct watering habits — distilled water, right timing, right level — is measured in dollars and minutes. The cost of not doing it is measured in premature battery replacement.
Frequently Asked Questions
Can I use bottled spring water for my golf cart batteries?
No. Spring water contains dissolved minerals — calcium, magnesium, and bicarbonates — that are specifically marketed as beneficial for drinking but are damaging to battery plates. Never use spring water, mineral water, or any flavored or enhanced water in golf cart batteries. Distilled water only.
How often should I check the water level in my golf cart batteries?
Every 30-45 days during regular use, or more frequently in hot weather (every 2-3 weeks above 85°F) and heavy use conditions. Always check after a full charge, never before. Set a recurring calendar reminder — this is the most commonly skipped maintenance task and the most impactful one.
What happens if I forget to add water and the plates get exposed?
Lead plates exposed to air oxidize rapidly, forming a layer of insoluble lead compounds on the exposed surface. This reduces the active surface area of the plate permanently—there is no treatment that reverses the oxidation. The sooner you notice and refill with distilled water, the less permanent damage results. Hours of exposure cause less damage than days; days cause less than weeks. If a cell has been dry for an extended period, have the battery load-tested to assess remaining capacity.
Is purified water the same as distilled water?
Not necessarily. ‘Purified’ is a broad marketing term that can describe water treated by any of several processes—filtration, RO, deionization, or distillation. Only distillation and deionization consistently produce the near-zero TDS levels required for battery use. Always check the TDS level with a meter if using anything labelled ‘purified’ rather than explicitly ‘distilled’ or ‘deionised.’
My battery is using a lot more water than usual. Why?
Significantly higher than normal water consumption is almost always caused by a charger that is not terminating correctly at the end of the charge cycle. A charger that overshoots the target absorption voltage or that does not switch off and continues pushing current into a fully charged pack electrolyzes water at a high rate. Have your charger’s output voltage tested during the absorption phase—it should be reaching 58.4-59.5 V on a 48 V system and then reducing current naturally as the pack approaches full charge. If the charger is running continuously without tapering, it is overcharging and needs repair or replacement.
Does the type of water matter if I have AGM or lithium batteries?
For AGM batteries — no, because AGM batteries are sealed and require no water addition. Their electrolyte is immobilized in the glass mat and is factory-sealed. For lithium batteries — no, they have no liquid electrolyte at all. The distilled water question applies only to flooded lead-acid batteries, which are the most common type in golf carts produced before approximately 2020.
I accidentally added tap water to my batteries. What should I do?
First, do not panic — a single instance of tap water will not cause immediate failure. Second, complete a full charge and equalization cycle immediately to mix the electrolyte thoroughly and minimize localized mineral concentration on the plates. Third, switch permanently to distilled water for all future fills. The contamination from a single tap water addition is small, and the long-term impact is modest if corrected promptly. Repeated tap water use over months and years is where significant damage accumulates.
The Bottom Line: Simple Discipline, Major Impact
Golf cart battery watering is the maintenance task with the highest impact per minute invested. The procedure takes five minutes monthly. The materials cost pennies per fill. And the difference in battery lifespan between a properly watered pack and a neglected one is measured in years and hundreds of dollars.
The three rules that govern everything in this guide are use distilled water, always fill after a full charge (never before), and fill to the correct level—below the filler tube, not to the top. These three rules, applied every 30-45 days, are the most powerful maintenance intervention available for any flooded lead-acid golf cart battery.
Everything else in battery maintenance — equalization, terminal cleaning, load testing — matters. But none of it compensates for watering with the wrong water or at the wrong time. The water chemistry is the foundation. Get it right from the first fill, and the rest of the maintenance program delivers its full benefit.